When placing dangerous substances or mixtures on the market the REACH Regulation and the Ordinance on Hazardous Substances request to protect health and environment on a necessary level. Information about the hazard of a chemical substance have to be provided independent from quantity as Safety Data Sheet (SDS) in the particular language.
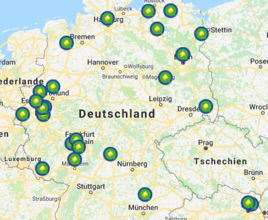
For several years now, AGQM successfully supports interested companies to provide and update their Safety Data Sheets.
Depending on the hazard of the used substance the SDS has to be provided unrequested (substance of very high concern – SVHC), upon request (hazardous to health or environment in low individual concentration) or optional (basis for hazard assessment) from the first day of delivery. SDS for substances subject to registration have to comply with the content of the REACH dossier for the substance. This applies explicit for planned applications and exclusion from applications.
From the Safety Data Sheet obligations result which producers, suppliers and users have to comply with.
The obligations include an immediate update of the SDS when new information are available that can have an impact on risk management measures. The SDS has to be updated as well when the legal situation has changed because of granting or rejection of an admission or limitation. If a chemical safety report was written, additional exposition scenarios have to be attached and an “extended SDS” has to be created.
For creation and updating of Safety Data Sheets, AGQM offers an Update and Care Contract.
The contract includes next to the compilation of SDS in different languages the monitoring of the legal situation and the annual incorporation of necessary updates and operational changes. Company related data necessary for the fulfilment of the contract will be treated as confidential.
You can find detailed information about the costs of the Update and Care Contract here.
If you are interested, you can contact us under ernpu@ntdz-ovbqvrfry.qr.